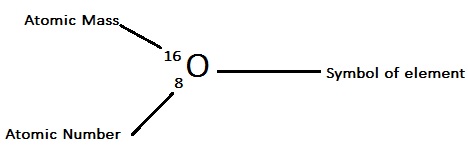
So, moles are used in chemical calculations. However, even though it is very difficult, scientists still need a unit of measurement to represent all these elementary entities. Therefore, representing such millions of atoms is quite impossible. Each chemical reaction involves an array of atoms that interact with each other differently under different conditions. This is because there can be millions of chemical reactions. This Avogadro’s number, along with mole, are very foundational concepts in any calculations in physical chemistry. Scientists call this Avogadro’s number after Avogadro’s name. The number of atoms present is 6.02214076 X 10^23. Therefore, one mole of carbon contains the same number of elementary entities as the number of atoms. Atoms and molecules are the smallest units of elements and compounds respectively. Scientists use the concept of moles to calculate the number of elements that are present in a particular sample of chemicals. A mole, therefore, is a unit that helps in measuring and stating the amount of matter a given substance contains. However, in the question of moles, the definition does not explain much. 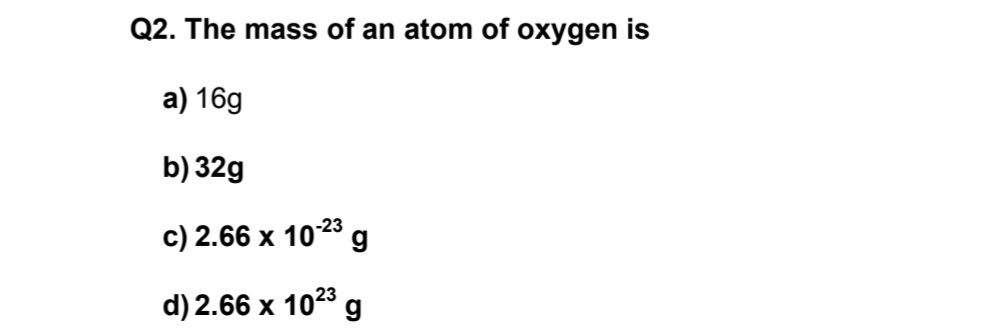
It is the mass one mole of that element or compound possesses. The definition of the molar mass of a chemical substance is a little complex. Its molar mass will be 117.10 grams per mole. So, Calcium hydrogen carbonate (Ca(HCO3)2) will have a formula mass of 117.10 amu. However, the term molar mass holds if we are considering a mole of the concerned ionic compound. This is because ionic compounds cannot dissociate into individual molecules. 
In such cases, we use the term “formula mass”. However, we can’t apply the term “molar mass” while considering non-molecular or ionic compounds. Therefore, by that logic, the molar mass of a N2 molecule is 28.02 grams per mole. What results from this is the molecular mass of the concerned molecule. The atomic mass of an element is the cumulative addition of all the atomic masses of the atoms it contains. Here 14 is the atomic mass of an individual Nitrogen atom. So, for NItrogen the atomic mass is 14.01 X 2 = 28.02 amu. For instance, nitrogen or N2 has a mass which is the total of the atomic masses of 2 individual nitrogen atoms- N. The molar mass of individual elements helps in calculating compounds as well. Consequently, we will realize its importance. So, for this, we need to know what molar mass is and how it is calculated while solving numericals in physical chemistry. Knowing the molar mass of oxygen is very important. Oxygen is one of the most abundant elements in the universe and is only next to nitrogen in the atmosphere.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
